Greek Philosophers
|
Around 450 B.C. the Greeks believed that all matter was composed of four basic elements. These were water, fire, air, or earth. The water was the blood in their bodies, the fire was their body temperature, air was their breath, and dirt was their bones. The Greeks believed this until Democritus later came along and proposed his theories.
|
Resources:
|
Democritus
|
Democritus' Atomic model was a simple sphere. He proposed that instead of the four elements, everything was made up of tiny particles called Atomos. His model would have changed the way the Greeks believed if not for Aristotle, who asked how the atoms stayed together. Democritus could not answer Aristotle's question, which as a result led to his ideas being rejected, but would later be brought up again and proven true by John Dalton.
John Dalton
|
Resources:
|
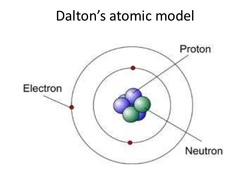
John Dalton came along and proved Democritus' theories to be true. He
also expanded upon Democritus' atomic model. Electrons and neutrons were unknown until John Dalton proposed his model. His model added these molecules to the ever changing atomic model. |
J . J . Thomson
|
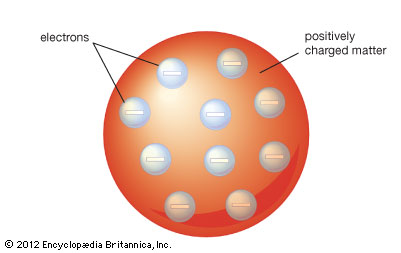
J.J. Thomson's atomic model, known as the plum pudding model, showed electrons embedded around in positively charged matter. In 1906, Thomson proposed that atoms had far fewer electrons, a number roughly equal to the atomic number. This was correct seeing as how we know how many electrons are in an element by its atomic number.
Resources:
|
Ernest Rutherford
Resources:
- http://www.britannica.com/science/Rutherford-atomic-model
- http://socratic.org/questions/how-did-rutherford-s-gold-foil-experiment-change-the-model-of-the-atom
- http://www.biography.com/people/ernest-rutherford-39099
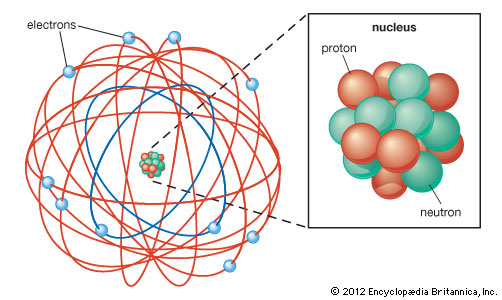
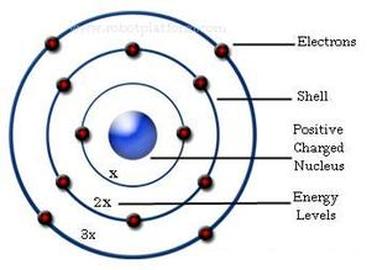
Ernest Rutherford created the Rutherford model, also known as the nuclear atom or planetary model of the atom. The model described the atom as a small, dense, positively charged core called a nucleus, which almost all the mass is concentrated, around which the light, negative constituents, called electrons, circulate at a distance. Rutherford calculated the size of the nucleus to be about 1/10,000 of the size of the atom. Rutherford replicated his model by using gold foil and releasing a radioactive material encased in lead at it. The positively charged particles were aimed at the gold foil and some were deflected straight back, some were deflected slightly, and some were not deflected at all. This proved Thomson's model of the atom to be incorrect.
James ChadwickResources:
https://sites.google.com/site/historyofanatom/james-chadwick http://www.tutorvista.com/science/chadwick-atomic-model James Chadwick, one of Ernest Rutherford's co-workers, who contributed to the atomic model by making the discovery of the neutron. He did this by using evidence from by Irene Joliot-Curie, who discovered that when beryllium was bombarded with positively charged alpha particles a beam with a high penetrating power was formed. Chadwick saw that this beam was not deflected by electric or magnetic fields, which meant that it contained neutral particles. His model was much the same as Rutherford's, except for the fact that he added neutrons.
|
Niels Bohr
|
Resources:
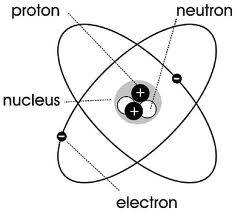
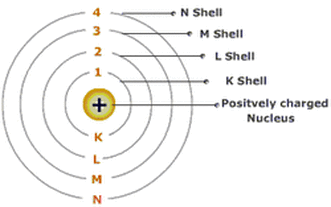
Like Rutherford, Niels Bohr's model had a densely packed nucleus. His model added shells also( N Shell, M Shell, L Shell, K Shell). Bohr expanded upon Rutherford's ideas by putting more attention on describing the electron. Bohr stated they moved in fixed, circular orbits around the nucleus. These were called electron shells. These shells were at set distances away from the nucleus and were the same for all atoms. He said that an electrons can go from a lower orbit to a higher orbit by absorbing energy, which can be done in the forms of heat, light or electricity.
|
Electron Cloud Model
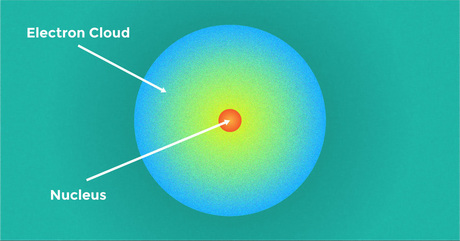
The Electron Cloud Model is the model where atoms are not shown as particles moving around the nucleus in a fixed orbit. Instead, the electrons are depicted as a “cloud”, representing their probable locations surrounding the nucleus, instead of distinct particles exactly positioned along an orbit. Heisenberg's Uncertainty Principle was a factor in this change, as it states that you can't know the precise location and momentum of a particle.
Resources:
Quantum Mechanical Model of the Atom
|
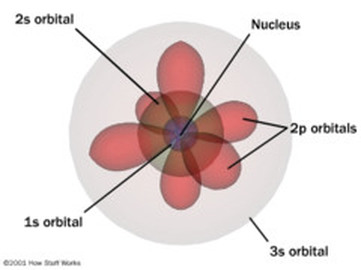
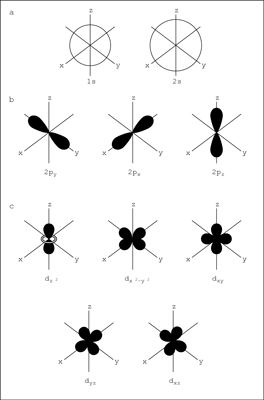
The Quantum Mechanical Model of the Atom shows how electrons exist in atoms, and how the electrons determine the chemical and physical properties of elements. This model is made by the Electro Configuration Sentence. It has different shapes like circles or infinity signs. The circles correspond to the 1s, 2s, 3s, and so on. The infinity shapes correspond to the 2p, 3p, and so forth. This model explains the current periodic tables, and provides the basis for our understanding of chemical bonding.
|