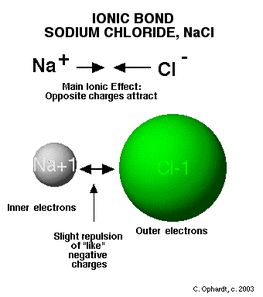
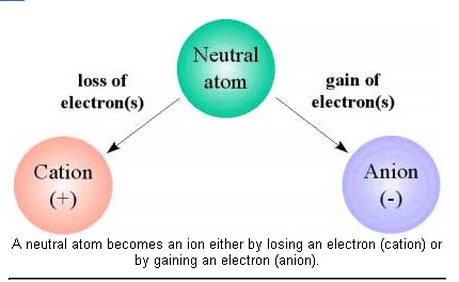
Ionic Bonds form through valence electrons exchanging between atoms, primarily a metal and a nonmetal.
https://www.boundless.com/chemistry/textbooks/boundless-chemistry-textbook/atoms-molecules-and-ions-2/types-of-chemical-bonds-36/ionic-bonds-207-1862/
https://www.boundless.com/chemistry/textbooks/boundless-chemistry-textbook/atoms-molecules-and-ions-2/types-of-chemical-bonds-36/ionic-bonds-207-1862/
|
The loss or gain of valence electrons lets the ions abide by the octet rule and become stable. The octet rule is when atoms are most stable when there are eight valence electrons in its valence shell. Ionic Compounds are usually neutral, so they combine in ways that neutralize there charge.
https://www.boundless.com/chemistry/textbooks/boundless-chemistry-textbook/atoms-molecules-and-ions-2/types-of-chemical-bonds-36/ionic-bonds-207-1862/ Ionic bonds are the result of the attraction between a positive and negative ions. In Crystalline Compounds, the net balance of forces is called Lattice Energy, which is the energy released in the formation of an ionic compound.
http://chemistry.elmhurst.edu/vchembook/143Aioniccpds.html |
Rules for Ion formation and naming
|
http://syllabus-msc.blogspot.com/2010/12/b-ionic-bond.html
|
What are Ions?
Ions are an atom or molecule with a net electric charge due to the loss or gain of one or more electrons.
How do Ions bond and why?
They transfer valence electrons between atoms so that they can become stable.
Metallic Bonding -
The characteristic of metals where mobile valence electrons are shared among atoms in a usually stable crystalline structure.
Electron Sea Model -
A model of metallic bonding in which cations are considered to be fixed points within a mobile "sea" of electrons.
Ions are an atom or molecule with a net electric charge due to the loss or gain of one or more electrons.
How do Ions bond and why?
They transfer valence electrons between atoms so that they can become stable.
Metallic Bonding -
The characteristic of metals where mobile valence electrons are shared among atoms in a usually stable crystalline structure.
Electron Sea Model -
A model of metallic bonding in which cations are considered to be fixed points within a mobile "sea" of electrons.